It’s kinda inevitable to repress the assimilation of technology into every sector of our life. The improvement of services provided by technology is becoming a lot easier and Omron see the future of the medical devices hinge on the addition of features based on technology.

Omron Healthcare has gotten FDA approval for its Complete™, pulse screen with EKG capacity. Complete is the principal pulse screen with EKG capacity in a solitary gadget.
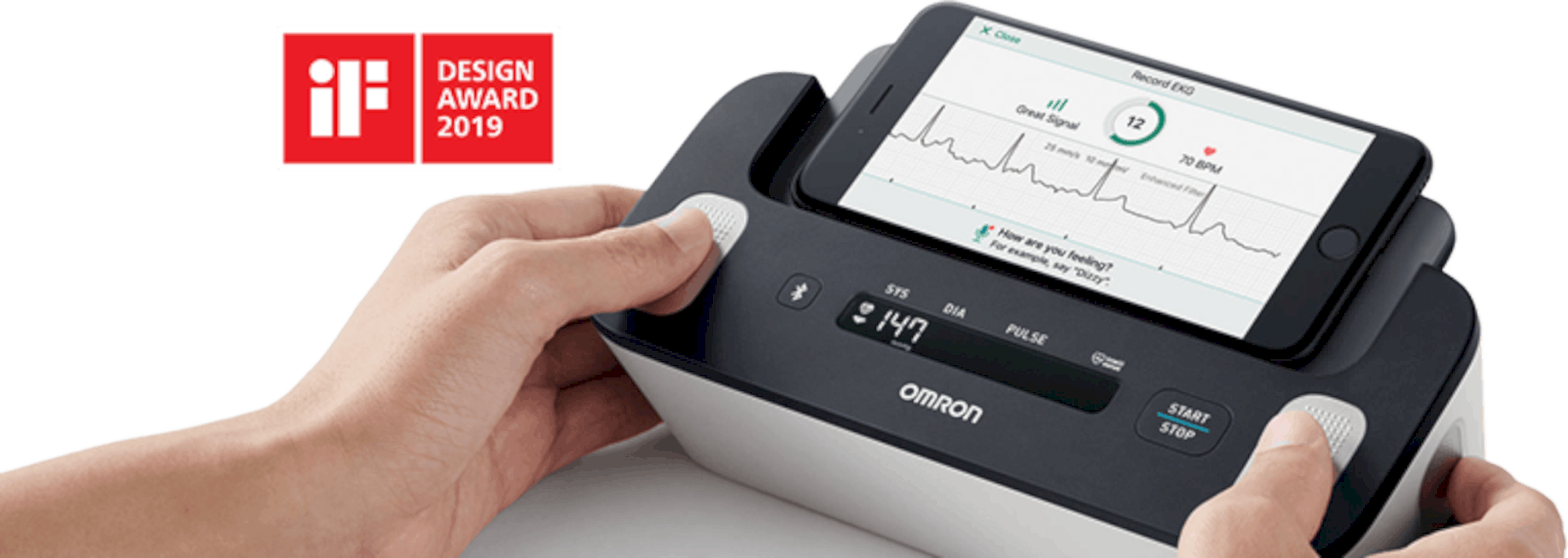
The gadget is an upper arm pulse screen that enables clients to at the same time screen EKG and circulatory strain readings at home. EKG readings can be estimated by contacting anodes strategically placed on the top face and the two sides of the screen for simple access and solace.
EKG is a fundamental estimation for those with atrial fibrillation, otherwise called arrhythmia or Afib, a condition causing unpredictable heartbeat that can prompt heart wellbeing related intricacies, including the expanded danger of stroke and heart failure.
Omron created Complete with collaboration with AliveCor, the market leader in FDA-cleared individual EKG innovation. Complete utilization of the progressed new calculation planned by AliveCor for improved recognition of the likelihood of AFib alongside believed medicinal evaluation circulatory strain estimation from Omron.

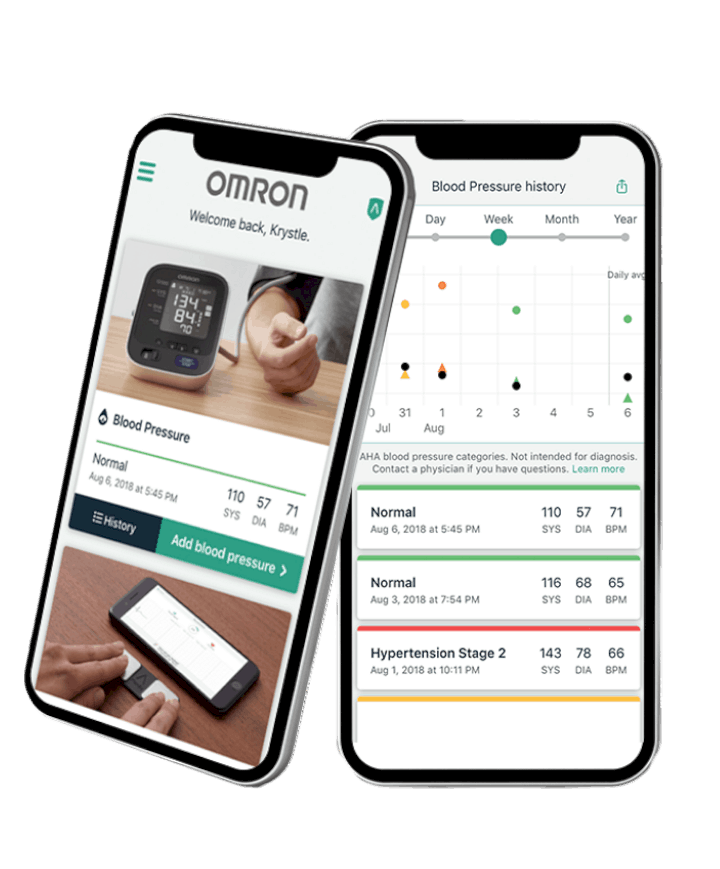
Complete associated with the exclusive Omron versatile application Omron Connect, which enables clients to store, track information after some time and offer their heart wellbeing information with their doctor.
This product as of late also won the iF Design Award 2019, introduced by iF International Forum Design GmbH, one of the world’s most renowned design awards.

Complete was made accessible for pre-deal on April 15 and was met with positive customer reaction and request. Omron is planning to release Complete in the U.S. for $199.99 and accessible on OmronHealthcare.com on May 1, 2019, and Amazon in the not so distant future.



